Laboratory of Naomi L. Sayre, Ph.D.
Postdoctoral Research Opportunity
The laboratory of Dr. Naomi Sayre is accepting applications for a T32 postdoctoral research fellow studying the effect of LDLR-like protein 1 (LRP1) on neural stem cell behavior after stroke. This research training program is designed to prepare early career scientists to become independent investigators who can work in teams to discover answers that will lead to the prevention and treatment of occlusive vascular disease. This NIH-sponsored NRSA postdoctoral research training program in cardiovascular pathobiology (HL07446) offers concentrated interdisciplinary research training across diverse laboratories with wide-ranging areas of expertise. Successful applicants will be highly motivated postdoctoral trainees interested in in vivo mouse models of disease, cell biology, and neuroscience.
All postdoctoral appointments are designated as security sensitive positions. The University of Texas Health Science Center at San Antonio is an Equal Employment Opportunity/Affirmative Action employer.
For more information on possible training opportunities, please contact sayre@uthscsa.edu.
Low-density lipoprotein receptor related protein 1 (LRP1)
LRP1 is an important plasma membrane receptor involved in the receptor mediated endocytosis of lipoproteins from cerebrospinal fluid. In addition, LRP1 binds to a range of molecules and proteins, both from the extracellular space and on the plasma membrane, thereby regulating their amount in either compartment. Such proteins include Apolipoprotein E, amyloid beta, tumor necrosis factor receptor, bone morphogenic protein signaling mediators, and multiple others. Altogether, we hypothesize that by modulating plasma membrane protein expression, LRP1 has the potential to significantly influence cellular response after brain damage. To this end, we utilize genetic mouse models to study the effect of LRP1 knockout specifically within astrocytes or neural stem cells, and the effect that cell-specific knockout might have on the brain response after damage.
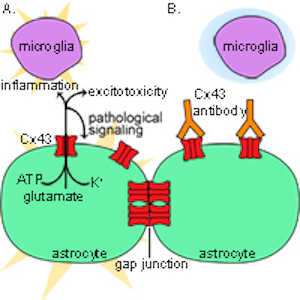
Connexin-43 function blocking antibodies for the treatment of spinal cord injury
In collaboration with Dr. Jean Jiang in the Department of Biochemistry, we are testing the effect of a function blocking antibody to Connexin-43 (Cx-43) hemichannels on damage after spinal cord injury. Our long-term goal is to improve the therapeutic potential of patients who suffer from traumatic spinal cord injury (SCI). Every year, thousands of patients are admitted to U.S. hospitals due to traumatic spinal injury.
Part of the post-injury neuroinflammatory process is the activation of astrocytes and formation of a glial scar resulting in an impermeable milieu for axonal regeneration. At the time of injury, astrocytic Cx43 hemichannels open, releasing a quantity of ATP, glutamate, ions and other molecules. The net result of pathological opening of the Cx43 hemichannels is an increase in metabolic stress, imbalanced ion homeostasis, excitotoxicty, and inflammation at the site of injury.
Altogether this causes spread of secondary damage after the initial injury. We hypothesize that inhibiting the function of Cx43 hemichannels after SCI will limit the size of spinal cord lesion after trauma and improves functional recovery. Recently, Dr. Jiang’s laboratory developed a potent antibody that blocks Cx43 hemichannels. Dr. Sayre’s laboratory developed a mouse model of SCI, which we are using to test the effect of Cx43 inhibition on SCI damage. We ultimately aim to utilize this antibody as an approved treatment for SCI.

