Lynette C. Daws, Ph.D.
Professor
Education
B.S. (Honors), Flinders University of South Australia, 1989
Ph.D., Flinders University of South Australia, 1994
Research
Research in the Daws lab focuses primarily on biogenic amine transporters, how they are involved in psychiatric disease states, such as depression and addiction, and importantly, how they are modulated by therapeutics, drugs of abuse, and novel drugs to treat these disorders. The Daws lab is well known for its use of in vivo electrochemical techniques, primarily chronoamperometry, biochemical and behavioral approaches to measure the activity of these transporters in living animals. We study high-affinity, low-capacity (“uptake 1”) transporters, which include serotonin, dopamine and norepinephrine transporters (SERT, DAT and NET), as well as low-affinity, high-capacity (“uptake 2”) transporters, including organic cation transporters (OCTs) and plasma membrane monoamine transporter (PMAT). Current research builds on our seminal findings that organic cation transporter 3 (OCT3) is an important contributor to clearance of extracellular serotonin, especially when SERT function is pharmacologically or genetically impaired. We are currently investigating how SERT and OCT3 regulation of extracellular serotonin influences neuronal responses to serotonin in mood relevant circuits, and the consequences for behavior. Our findings make OCT3 an attractive target for development of novel therapeutics to treat a host of psychiatric disorders, especially those where current medications (e.g. selective serotonin reuptake inhibitors, such as Prozac) are suboptimal, or ineffective, in providing symptom relief.
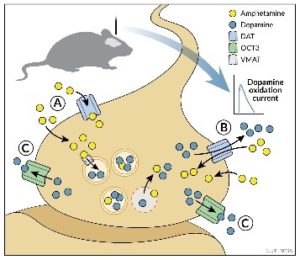
Another major area of research builds on our findings that several drugs of abuse, including amphetamine and mephedrone, produce their effects, in part, via OCT3. We are currently investigating OCT3 as a putative target for the development of novel therapeutics for treatment of addiction to these, and related drugs of abuse. Other ongoing research includes investigation of insulin regulation of DAT in eating disorders, studies of regulation of monoamine transporters by auto- and hetero-receptors, and collaborations with medicinal chemists to develop drugs selective for OCT3. The Daws lab provides a dynamic and productive working environment. Members currently include Dr. Georgianna Gould (Associate Professor/Research); W. Anthony Owens (Research Scientist), Rebecca Horton (Research Associate), Robert Hammack (Graduate Student), Lauren Honan (Graduate Student) and Dr. Nikki Clauss (Post-Doctoral Fellow). Key Words: Organic Cation Transporters; Plasma Membrane Monoamine Transporter; Serotonin Transporter; Dopamine Transporter; Norepinephrine Transporter, Depression; Stress; Substance Use Disorders; Addiction; Eating Disorders; In Vivo Chronoamperometry; Behavior
Lab Members
 | W. Anthony Owens, MS Research Scientist |
 | Samuel Swinford Senior Research Assistant |
 | Gopakumar Changarathil, Ph.D. Postdoctoral Fellow |
 | Colby E. Witt Ph.D. Postdoctoral Fellow |
 | Yibin “Clara” Xu, PhD Postdoctoral Fellow |
 | Simon Tangen Graduate Student |
Publications
| Mayer, F.P., Stewart, A., Varman, D.R., Moritz, A.E., Foster, J.D., Owens, A.W., Areal, L.B., Gowrishankar, R., Velez, M., Wickham, K., Phelps, H., Katamish, R., Rabil, M., Jayanthi, L.D., Vaughan, R., Daws, L.C., Blakely, R.D., Ramamoorthy, S. (in press) Kappa opioid receptor antagonism rescues genetic perturbation of dopamine homeostasis: Molecular, physiological and behavioral consequences. Preprint in BioRxiv, DOI: 10.1101/2023.05.03.539310, Molecular Psychiatry |
| Berg, K.A., Clarke, W.P., Daws, L.C., Frazer, A., Girotti, M. and Lodge, D.J. (in press) Neurotransmitters, receptors and transporters in psychiatric disorders. Chapter 2 In The American Psychiatric Association Publishing Textbook of Psychopharmacology, Sixth Edition; Editors Alan F. Schatzberg, M.D., and Charles B. Nemeroff, M.D., Ph.D. |
| Clauss, N.J., Mayer, F.P., Owens, W.A., Vitela, M., Clarke, K.M., Bowman, M.A., Horton, R.E., Gründemann, D., Schmid, D., Holy, M., Gould, G.G., Koek, W., Sitte, H.H., and Daws, L.C. (2023) Ethanol inhibits dopamine uptake via organic cation transporter 3: Implications for ethanol and cocaine co-abuse. Molecular Psychiatry. Jun 13. doi: 10.1038/s41380-023-02064-5. Online ahead of print. |
| Bowman, M.A., Gomez, J.A., Mitchell, N.C., Wells, A.M., Vitela, M., Clarke, K.M., Horton, R.E., Koek, W., Daws, L.C. (2022) Faster serotonin clearance in CA3 region of hippocampus and antidepressant-like effect of decynium-22 in juvenile mice are putatively linked to increased plasma membrane monoamine transporter function: Implications for efficacy of antidepressants in juveniles. Cells, section “Cell Signaling”, special issue on Transporters in Health and Disease, 11(15):2454, doi: 10.3390/cells11152454. |
| Gould, G.G., Barba-Escobedo, P., Horton, R.E. and Daws, L.C. (2022) High affinity brain membrane homogenate binding to and reduced dorsal camouflaging after acute decynium-22 exposures in zebrafish. Frontiers in Pharmacology, section Experimental Pharmacology and Drug Discovery un 9;13:841423. doi: 10.3389/fphar.2022.841423. eCollection 2022. |
| Clauss, N.J., Koek, W. and Daws, L.C. (2021) Role of organic cation transporter 3 and plasma membrane monoamine transporter in the rewarding properties and locomotor sensitizing effects of amphetamine in male and female mice. International Journal of Molecular Sciences 22(24):13420. doi: 10.3390/ijms222413420. This article belongs to the Special Issue Physiology, Biochemistry, and Pharmacology of Transporters for Organic Cations 2.0 |
| Clauss, N.J. and Daws, L.C. (2021) Neurotransmitter transporters and their role in the pharmacological actions of therapeutic and abused drugs. Comprehensive Pharmacology, In: Kenakin, T. (Ed.), Comprehensive Pharmacology. vol. 1, pp. 165–204. Elsevier. https://dx.doi.org/10.1016/B978-0-12-820472-6.00172-9. |
| Daws LC. (2021) Organic Cation Transporters in Psychiatric Disorders. Handb Exp Pharmacol. 2021 Jul 20. doi: 10.1007/164_2021_473. Online ahead of print. PMID: 34282486 |
| Maier J, Niello M, Rudin D, Daws LC, Sitte HH. (2021) The Interaction of Organic Cation Transporters 1-3 and PMAT with Psychoactive Substances. Handb Exp Pharmacol. 2021 May 17. doi: 10.1007/164_2021_469. Online ahead of print. PMID: 33993413 |
| Bowman, M.A., Vitela, M., Clarke, K.M., Koek, W. and Daws, L.C. (2020) Serotonin transporter and plasma membrane monoamine transporter are necessary for the antidepressant-like effects of ketamine in mice. International Journal of Molecular Sciences Oct 14;21(20):7581. doi: 10.3390/ijms21207581. PMID: 33066466 |
| Bowman, M.A., Mitchell, N.C., Owens, W.A., Horton, R.E., Koek, W. and Daws, L.C. (2020) Effect of concurrent organic cation transporter blockade on norepinephrine clearance inhibiting- and antidepressant-like actions of desipramine and venlafaxine. European Journal of Pharmacology Sep 15;883:173285. doi: 10.1016/j.ejphar.2020.173285. Epub 2020 Jul 19. PMID: 32697958 |
| Jensen, M.E., Galli, A., Thomsen, M., Jensen, K.L., Thomsen, G.K., Klausen, M.K.K., Vilsbøll, T., Christensen, M.B., Holst, J.J., Owens, W.A., Robertson, S., Daws, L.C., Zanella, D., Gether, U., Knudsen, G.M. and Fink-Jensen, A. (2020) Glucagon-like peptide-1 receptor regulation of basal dopamine transporter activity is species dependent. Neurochemistry International Sep;138:104772. doi: 10.1016/j.neuint.2020.104772. Epub 2020 May 25. PMID: 32464226 |
| Annamalai, B., Ragu Varman, D., Horton, R.E., Daws, L.C., Jayanthi, L.D. and Ramamoorthy, S. (2020) Histamine receptors regulate the activity, surface expression and phosphorylation of serotonin transporters. ACS Chemical Neuroscience Epub ahead of print |
| Gilman, T.L., George, C.M., Andrade, M., Mitchell, N.C., Toney, G.M. and Daws, L.C. (2019) High salt intake lowers behavioral inhibition. Frontiers in Behavioral Neuroscience 13:271 ePub |
| Gilman, T.L., Owens, W.A., George, C.M., Metzel, L., Vitela, M., Ferreira, L., Bowman, M.A., Gould, G.G., Toney, G.M., Daws, L.C. (2019) Age- and sex-specific plasticity in dopamine transporter function revealed by food restriction and exercise in a rat activity-based anorexia paradigm. Journal of Pharmacology and Experimental Therapeutics 371(2):268-277 |
| Garbarino, V.R., Santos, T.A., Nelson, A.R., Daws, L.C., Gould, G.G. (2019) Prenatal metformin exposure curbs adolescent male social interaction preference. Pharmacological Research 140:21-32. Epub 2018 PMID:30423430 |
| Gilman, T.L., Mitchell, N.C., Daws, L.C., Toney, G.M. (2019) Neuroinflammation contributes to high salt intake-augmented neuronal activation and active coping responses to an acute stressor. International Journal of Neuropsychopharmacology 22(2):137-142 PMCID:PMC6368371 |
| Fraser-Spears, R., Krause-Heuer, A.M., Basiouny, M., Mayer, F.P., Manishimwe, R., Wyatt, N.A., Dobrowolksi, J.C., Roberts, M.P., Greguric, I., Kumar, N., Koek, W., Sitte, H.H., Callaghan, P.D., Fraser, B.H., Daws, L.C. (2019) Comparative analysis of novel decynium-22 analogs to inhibit transport by the low-affinity, high-capacity biogenic amine transporters, organic cation transporters 2 and 3, and plasma membrane monoamine transporter. European Journal of Pharmacology 842:351-364. Epub 2018 PMID:30473490 |
| Mayer, F.P., Schmid, D., Holy, M., Daws, L.C., Sitte, H.H. (2019) Polytox” synthetic cathinone abuse: a potential role for organic cation transporter 3. Neurochemistry International 123:7-12. [Epub ahead of print] PMID:30248432 |
| Garbarino, V.R., Gilman, T.G., Daws, L.C., and Gould, G.G. (2019) Extreme enhancement or depletion of serotonin transporter function and serotonin availability in autism spectrum disorder. Pharmacol Res. 140:85-99. doi: 10.1016/j.phrs.2018.07.010. Epub 2018 Jul 24. Review.. PMID:30009933 |
| Bowman, M.A., and Daws, L.C. (2019) Targeting serotonin transporters in the treatment of juvenile and adolescent depression. Frontiers in Neuroscience, Section Neuropharmacology, Special Issue, Serotonin, receptors and transporters: Exploring new and known signaling pathways to improve the efficacy of antidepressant treatment. 13:156. doi: 10.3389/fnins.2019.00156. eCollection 2019. PMCID:PMC6401641 |
| Gilman, T.L., George, C.M., Vitela, M., Herrera-Rosales, M., Basiouny, M., Koek, W., Daws, L.C. (2018) Constitutive plasma membrane monoamine transporter (PMAT, Slc29a4) deficiency subtly affects anxiety-like and coping behaviors. European Journal of Neuroscience May 24. doi: 10.1111/ejn.13968. [Epub ahead of print] PMID:29797618 |
| Jensen, K.L., Sørensen, G., Dencker, D., Owens, W.A., Rahbek-Clemmensen, T., Lever, M.B., Runegaard, A.H., Christensen, N.R., Weikop, P., Wörtwein, G., Fink-Jensen, A., Madsen, K.L., Daws, L.C., Gether, U., Rickhag, M. (2018) Attenuated behavioral responses to cocaine in mice lacking PICK1 is associated with dysregulation of dopamine homeostasis. eNeuro 5(3). pii: ENEURO.0422-17 |
| Mitchell, N.C., Gilman, T.L., Daws, L.C., Toney, G.M. (2018) High salt intake enhances swim stress-induced PVN vasopressin cell activation and active stress coping. Neuropsychoendocrinology 93:29-38 |
| Mayer, F.P., Schmid, D., Owens, W.A., Gould, G.G., Salzer, I., Böhm, S., Chiba, P., Williams, P., Apuschkin- Christiansen, M., Wu, H-H., Gether, U., Koek, W., Daws, L.C*., Sitte, H.H.* (2018) An unsuspected role for organic cation transporter 3 in the actions of amphetamine. Neuropsychopharmacology 43(12):2408-2417. * co-corresponding authors, contributed equally |
| Daws, L.C., Owens, W.A. and Toney, G.M. (2016) High-speed chronoamperometry to measure biogenic amine release and uptake in vivo. In: Neurotransmitter Transporters – Investigative Methods, (Eds. Harald Sitte and Heinz Bonisch). Humana Press, pp 53-81. |
|
Gasser, P.J., Daws, L.C. (2017) Extending the family: Roles for uptake2 transporters in regulation of monoaminergic signaling. Chemical Neuroanatomy ePub ahead of print, 2017 Oct;83-84:107-108. doi: 10.1016/j.jchemneu.2017.07.009. Epub 2017 Jul 28. No abstract available.
|
|
Krause-Heuer, A.M.,† Fraser-Spears, R.†, Dobrowolski, J., Ashford, M.E., Wyatt, N.A., Roberts, M.P., Gould, G.G., Cheah, W-C., Ng, C., Bhadbhade, M., Zhang, B., Greguric, I., Wheate, N.J., Kumar, N., Koek, W., Callaghan, P.D., Daws, L.C*., Fraser, B.H.* (2017) Evaluation of the antidepressant therapeutic potential of isocyanine and pseudoisocyanine analogues of the organic cation decynium-22. European Journal of Medicinal Chemistry 137:476-487. * co-corresponding authors, contributed equally; † co-first authors
|
|
Mitchell, N.C., Bowman, M.A., Gould, G.G., Koek, W., Daws, L.C. (2017) Ontogeny of NET expression and antidepressant-like response to desipramine in wild-type and SERT mutant mice. Journal of Pharmacology and Experimental Therapeutics 360(1):84-94.
|
|
Koek, W., Sandoval, T.L., Daws, L.C. (2018) Effects of the antidepressants desipramine and fluvoxamine on latency to immobility and duration of immobility in the forced swim test in adult male C57BL/6J mice. Behavioural Pharmacology 29(5):453-456.
|
|
Sung, U., Savchenko V., Binda F., Owens, W.A., Daws, L.C. (2017) Ca2+ dependent surface trafficking of norepinephrine transporters depends on threonine 30 and Ca2+ calmodulin kinases. Special Issue: Neurotransmitter Transporters in Health and Disease; Journal of Chemical Neuroanatomy, Epub ahead of print. Journal Cover
|
|
Sambo, D., Lin, M., Owens, W.A., Richardson, B., Jagnarine, D.A., Madhur, S., Alonge, T., Ali, M., Katz, J., Febo, M., Henry, K., Bruijnzeel, A.W., Daws, L.C., Khoshbouei, K. (2017) Sigma-1 receptor regulation of dopamine neurotransmission: beyond the surface membrane. Nature Communications 8(1):2228. doi: 10.1038/s41467-017-02087-x
|
|
Koek, W., Mitchell, N.C., Daws, L.C. (2018) Biphasic effects of selective serotonin reuptake inhibitors on anxiety: rapid reversal of escitalopram’s anxiogenic effects in the novelty-induced hypophagia (NIH) test in mice? Behavioural Pharmacology 29(4):365-369]
|
|
Mitchell, N.C., Owens, W.A.,Vitela, M., Gould, G., Koek, W.,Daws, L.C. (2016) Ontogeny of SERT expression and antidepressant-like response to escitalopram in wild-type and SERT mutant mice. Journal of Pharmacology and Experimental Therapeutics 358(2):271-281.
|
|
Mitchell, N.C., Koek, W., Daws, L.C. (2015) Antidepressant-like effects and basal immobility depend on age and serotonin transporter genotype. Genes, Brain and Behavior 14(7):543-549
|
|
Zhang, W.Q., Smolik, C.M., Barba-Escobedo, P.A., Gamez. M., Sanchez, J.J., Javors, M.A., Daws, L.C., Gould, G.G. (2015) Acute dietary tryptophan manipulation differentially alters social behavior, brain serotonin and plasma corticosterone in three iInbred mouse strains. Neuropharmacology Mar;90:1-8. doi: 10.1016/j.neuropharm.2014.10.024. Epub 2014 Nov 4.
|
|
Baladi, M.G., Daws, L.C., France, C.P. (2015) The impact of eating high fat chow on sensitivity to the locomotor stimulating effects of cocaine and on dopamine clearance in adolescent and adult male rats. International Journal of Neuropsychopharmacology Mar 24; 18(7):pyv024. doi: 10.1093/ijnp/pyv024.
|
|
Montañez, S., Munn, J.L., Owens, W.A., Horton, R.E., Daws, L.C. (2014) 5-HT1B receptor modulation of the serotonin transporter in vivo: Studies using KO mice. Neurochemistry International, (Special Issue: The Brain in Flux) 73:127-131.
|
|
Lamb, R.J., Pinkston, J.W., Daws, L.C. (2014) Ethanol effects on multiple fixed-interval, fixed-ratio responding in mice with deletions of the serotonin transporter gene. Behavioral Pharmacology 25(1):92-95
|
|
Decynium-22 enhances SSRI-induced antidepressant-like effects in mice: uncovering novel targets to treat depression. Horton RE, Apple DM, Owens WA, Baganz NL, Cano S, Mitchell NC, Vitela M, Gould GG, Koek W, Daws LC. J Neurosci. 2013 Jun 19;33(25):10534-43. doi: 10.1523/JNEUROSCI.5687-11.2013. PMID: 23785165 [PubMed – indexed for MEDLINE]
|
|
Membrane permeable C-terminal dopamine transporter peptides attenuate amphetamine-evoked dopamine release. Rickhag M, Owens WA, Winkler MT, Strandfelt KN, Rathje M, Sørensen G, Andresen B, Madsen KL, Jørgensen TN, Wörthwein G, Woldbye DP, Sitte H, Daws LC, Gether U.J Biol Chem. 2013 Jul 24. [Epub ahead of print] PMID:23884410[PubMed – as supplied by publisher]
|
|
Ethanol Self-Administration in Serotonin Transporter Knockout Mice: Unconstrained Demand & Elasticity. Lamb RJ, Daws LC. Genes Brain Behav. 2013 Aug 8. doi: 10.1111/gbb.12068. [Epub ahead of print] PMID: 23927813[PubMed – as supplied by publisher]
|
|
Lamb, R.J., Daws, L.C. (2013) Ethanol self-administration in serotonin knockout mice: Unconstrained demand and elasticity. Genes, Brain and Behavior 12(7):741-747.
|
|
Mitchell, N.C., Gould, G.G., Smolik, C.M., Koek, W., Daws, L.C. (2013) Antidepressant-like drug effects in juvenile and adolescent mice in the tail suspension test: Relationship with hippocampal serotonin and norepinephrine transporter expression and function. Frontiers in Pharmacology (Neuropharmacology), Special Issue: New frontiers in the neuropsychopharmacology of mental illness. [Epub ahead of print] Oct 28;4:131. doi: 10.3389/fphar.2013.00131. eCollection 2013.
|
|
Rickhag, M., Owens, W.A., Winkler M-T., Nørgaard-Strandfelt, K., Andresen, B., Sørenesn, G., Madsen, K.L., Wörtwein, G., Woldbye, D., Sitte, H.H., Daws, L.C., Gether, U. (2013) Membrane permeable C-terminal dopamine transporter peptides attenuate amphetamine-evoked dopamine release. Journal of Biological Chemistry 288(38):27534-27544. *selected as “Paper of the Week”.
|
|
Revisiting serotonin reuptake inhibitors and the therapeutic potential of “uptake-2” in psychiatric disorders. Daws LC, Koek W, Mitchell NC. ACS Chem Neurosci. 2013 Jan 16;4(1):16-21. doi: 10.1021/cn3001872. PMID:23336039[PubMed – indexed for MEDLINE]
|
|
Decynium-22 enhances SSRI-induced antidepressant-like effects in mice: uncovering novel targets to treat depression. Horton RE, Apple DM, Owens WA, Baganz NL, Cano S, Mitchell NC, Vitela M, Gould GG, Koek W, Daws LC. J Neurosci. 2013 Jun 19;33(25):10534-43. doi: 10.1523/JNEUROSCI.5687-11.2013. PMID: 23785165[PubMed – indexed for MEDLINE]
|
|
What’s old is new. Andrews AM, Daws LC. ACS Chem Neurosci. 2013 Jan 16;4(1):1-2. doi: 10.1021/cn300229z. No abstract available. PMID: 23336035 [PubMed – indexed for MEDLINE]
|
|
Revisiting serotonin reuptake inhibitors and the therapeutic potential of “uptake-2” in psychiatric disorders. Daws LC, Koek W, Mitchell NC. ACS Chem Neurosci. 2013 Jan 16;4(1):16-21. doi: 10.1021/cn3001872. PMID: 23336039 [PubMed – indexed for MEDLINE]
|
|
Inhibition of dopamine transporter activity by G protein βγ subunits.Garcia-Olivares J, Torres-Salazar D, Owens WA, Baust T, Siderovski DP, Amara SG, Zhu J, Daws LC, Torres GE. PLoS One. 2013;8(3):e59788. doi: 10.1371/journal.pone.0059788. Epub 2013 Mar 26. PMID: 23555781 [PubMed – in process]
|
|
Owens WA, Williams JM, Saunders C, Avison MJ, Galli A, Daws LC. Rescue of Dopamine Transporter Function in Hypoinsulinemic Rats by a D2 Receptor-ERK-Dependent Mechanism. J Neurosci. 2012 Feb 22;32(8):2637-47
|
|
You are what you eat: influence of type and amount of food consumed on central dopamine systems and the behavioral effects of direct- and indirect-acting dopamine receptor agonists. Baladi MG, Daws LC, France CP. Neuropharmacology. 2012 Jul;63(1):76-86. doi: 10.1016/j.neuropharm.2012.02.005. Epub 2012 Feb 23. Review.PMID: 22710441 [PubMed – indexed for MEDLINE]
|
|
Gould G.G., Hensler, J.G., Burke, T.F., Benno, R., Onaivi, E.S. and Daws, L.C. (2011) Density and Function of Central Serotonin (5-HT) Transporters, 5-HT1A and 5-HT2A Receptors, and Effects of their Targeting on BTBR T+tf/J Mouse Social Behavior. Journal of Neurochemistry 116(2):291-303.
|
|
Niswender, K., Daws, L.C., Avison, M.J. and Galli, A. (2011) Insulin regulation of monoamine signaling: Pathway to obesity. Neuropsychopharmacology 36:359-360.
|
|
Thompson, B.J., Jessen, T., Henry, L.K., Field, J.R., Gamble, K.L., Gresch, P.J., Carneiro, A.M., Horton, R.E., Chisnell, P.J., McMahon, D.G., Daws, L.C. and Blakely, R.D. (2011) Transgenic elimination of high-affinity antidepressant and cocaine sensitivity in the presynaptic serotonin transporter.Proceedings of the National Academy of Science 108(9):3785-3790
|
|
Daws, L.C., Avison, M.J., Robertson, S.D., Niswender, K., Galli, A. and Saunders, C. (2011) Insulin signaling and addiction. Neuropharmacology (Special issue on “Synaptic Plasticity and Addiction”) 61(7):1123-1128.
|
|
Daws, L.C. and Gould, G.G. (2011) Ontogeny and regulation of the serotonin transporter: Providing insights into human disorders. Pharmacology and Therapeutics 131(1):61-79.
|
|
Siuta, M., Robertson, S., Kocalis, H., Saunders, C., Gresch, P., Khatri, V., Shiota, C., Kennedy, J.P., Lindsley, C., Daws, L.C., Polley, D., Veenstra-Vanderweele, J., Stanwood, G., Magnuson, M., Niswender, K. and Galli, A. (2010) Dysregulation of the norepinephrine transporter sustains cortical hypodopaminergia and schizophrenia-like behaviors in neuronal Rictor null mice. Public Library of Science Biology 8(6):e1000393.
|
|
Robertson, S.D., Matthies, H.J.G., Owens, W.A., Sathananthan, V., Bibus, N.S., Kennedy, J.P., Lindsley, C.W., Daws, L.C.* and Galli, A.* (2010) Insulin signaling regulation of norepinephrine transporter (NET) surface availability and function, reveals Akt as a novel and potent regulator of the transporter. Journal of Neuroscience 30(34):11305-11316. * contributed equally, co-corresponding authors **Comment in “Letters to the Editor” Lang, U.E., Hortnagl, H., Lang, F. Modulation of norepinephrine brain concentrations through phosphoinositide 3-kinase siganling. Journal of Neuroscience Sept 22, 2011
|
|
Zhu, C-B., Lindler, K.M., Owens, W.A., Daws, L.C., Blakely, R.D. and Hewlett, W.A. (2010) IL-1R activation by systemic lipopolysaccharide induces behavioral despair linked to MAPK regulation of CNS serotonin transporters. Neuropsychopharmacology 35(13):2510-2520.
|
|
Baganz, N.L., Horton, R.E., Martin, K.P., Holmes, A. and Daws, L.C. (2010) Repeated swim impairs serotonin clearance via a corticosterone-sensitive mechanism: Organic cation transporter 3, the smoking gun. Journal of Neuroscience 30(45): 15185-15195.
|
|
Daws LC. Unfaithful neurotransmitter transporters: focus on serotonin uptake and implications for antidepressant efficacy. Pharmacol Ther. 2009 Jan;121(1):89-99.
|
|
France CP, Li JX, Owens WA, Koek W, Toney GM, Daws LC. Reduced effectiveness of escitalopram in the forced swimming test is associated with increased serotonin clearance rate in food-restricted rats.Int J Neuropsychopharmacol. 2009 Jul;12(6):731-6.
|
|
Sevak RJ, Koek W, Daws LC, Owens WA, Galli A, France CP. Behavioral effects of amphetamine in streptozotocin-treated rats. Eur J Pharmacol. 2008 Feb 26;581(1-2):105-12.
|
|
Sevak RJ, Koek W, Owens WA, Galli A, Daws LC, France CP. Feeding conditions differentially affect the neurochemical and behavioral effects of dopaminergic drugs in male rats. Eur J Pharmacol. 2008 Sep 11;592(1-3):109-15.
|
|
Baganz NL, Horton RE, Calderon AS, Owens WA, Munn JL, Watts LT, Koldzic-Zivanovic N, Jeske NA, Koek W, Toney GM, Daws LC. Organic cation transporter 3: Keeping the brake on extracellular serotonin in serotonin-transporter-deficient mice. Proc Natl Acad Sci U S A. 2008 Dec 2;105(48):18976-81.
|
|
Callaghan PD, Owens WA, Javors MA, Sanchez TA, Jones DJ, Irvine RJ, Daws LC. In vivo analysis of serotonin clearance in rat hippocampus reveals that repeated administration of p-methoxyamphetamine (PMA), but not 3,4-methylenedioxymethamphetamine (MDMA), leads to long-lasting deficits in serotonin transporter function. J Neurochem. 2007 Feb;100(3):617-27.
|
|
Sevak RJ, Owens WA, Koek W, Galli A, Daws LC, France CP. Evidence for D2 receptor mediation of amphetamine-induced normalization of locomotion and dopamine transporter function in hypoinsulinemic rats. J Neurochem. 2007 Apr;101(1):151-9.
|
|
Price DA, Owens WA, Gould GG, Frazer A, Roberts JL, Daws LC, Giuffrida A. CB1-independent inhibition of dopamine transporter activity by cannabinoids in mouse dorsal striatum. J Neurochem. 2007 Apr;101(2):389-96.
|
|
Daws LC, Munn JL, Valdez MF, Frosto-Burke T, Hensler JG. Serotonin transporter function, but not expression, is dependent on brain-derived neurotrophic factor (BDNF): in vivo studies in BDNF-deficient mice. J Neurochem. 2007 May;101(3):641-51.
|
|
Wiedholz L, Owens WA, Horton RE, Feyder M, Hefner KM, Sprengel R, Daws LC, Holmes A.Mice lacking the AMPA GluR1 receptor exhibit striatal hyperdopaminergia and ‘schizophrenia-related’ behaviors. Mol Psychiatry. 2007 Aug 7.
|
|
Williams JM, Owens WA, Turner GH, Saunders C, Dipace C, Blakely RD, France CP, Gore J.C., Daws LC, Avison MJ, Galli A. Hypoinsulinemia regulates amphetamine-induced reverse transport of dopamine. PLoS Biol. 2007 Oct 16;5(10):2369-78.
|
|
Daws, L.C., Montañez, S., Munn, J.L., Owens, W.A., Baganz, N.L., Boyce-Rustay J.M., Millstein, R., Wiedholz, L.M., Murphy, D.L. and Holmes, A. Ethanol inhibits clearance of brain serotonin by a serotonin transporter-independent mechanism. J Neurosci. 2006 Jun 14;26(24):6431-8.
|
|
Fog JU, Khoshbouei H, Holy M, Owens WA, Vaegter CB, Sen N, Nikandrova Y, Bowton E, McMahon DG, Colbran RJ, Daws LC, Sitte HH, Javitch JA, Galli A, Gether U. Calmodulin kinase II interacts with the dopamine transporter C terminus to regulate amphetamine-induced reverse transport. Neuron. 2006 Aug 17;51(4):417-29.
|
|
Callaghan PD, Farrand K, Salem A, Hughes P, Daws LC, Irvine RJ. Repeated administration of the substituted amphetamine p-methoxyamphetamine produces reductions in cortical 5-HT transporter binding but not 5-HT content, unlike 3,4-methylenedioxyamethamphetamine. Eur J Pharmacol. 2006 Sep 28;546(1-3):74-81.
|
|
Boyce-Rustay JM, Wiedholz LM, Millstein RA, Carroll J, Murphy DL, Daws LC, Holmes A. Ethanol-related behaviors in serotonin transporter knockout mice. Alcohol Clin Exp Res. 2006 Dec;30(12):1957-65.
|
|
Toney GM, Daws LC. Juxtacellular labeling and chemical phenotyping of extracellularly recorded neurons in vivo. Methods Mol Biol. 2006;337:127-37.
|
|
Daws, L.C. , Montañez, S., Owens, W.A., Gould G.G., Frazer, A., Toney, G.M. and Gerhardt, G.A. (2005) Transport mechanisms governing serotonin clearance in vivo revealed by high speed chronoamperometry. Journal of Neuroscience Methods – Special Issue, Studying monoamine transporters: Beyond hypermonoamineaemia. 143: 49-62.
|
|
Callaghan, P.D., Irvine, R.J. and Daws, L.C. Differences in the in vivo dynamics of neurotransmitter release and serotonin uptake after acute para-methoxyamphetamine and 3,4-methylenedioxymethamphetamine revealed by chronoamperometry. Neurochem Int. 2005 Oct;47(5):350-61.
|
|
Owens, W.A., Sevak, R., Galici, R., Chang, X., Javors, M.A., Galli, A., France, C.P. and Daws, L.C.Deficits in dopamine clearance and locomotion in hypoinsulinemic rats unmask novel modulation of dopamine transporters by amphetamine. J Neurochem. 2005 Sep;94(5):1402-10.
|
|
Ansah, T.A., Ramamoorthy, S., Montañez, S., Daws, L.C. and Blakely, R.D. (2003) Calcium-dependent inhibition of synaptosomal serotonin transport by the alpha 2-adrenergic receptor agonist UK14304. Journal of Pharmacology and Experimental Therapeutics 305:956-965.
|
|
Montañez, S., Owens, W.A., Gould, G.G., Murphy, D.L. and Daws, L.C. (2003) Exaggerated effects of fluvoxamine in heterozygote serotonin transporter knockout mice. Journal of Neurochemistry 86:210-219.
|
|
Daws, L.C. , Callaghan, P.D., Morón, J., Kahlig, M.K., Shippenberg, T.S., Javitch, J.A. and Galli, A. (2002) Cocaine increases dopamine uptake and cell surface expression of dopamine transporters.Biochemical and Biophysical Research Communications 290(5):1545-1550.
|
|
Daws, L.C. , Irvine, R.J., Callaghan, P.D., Toop, N.P., White, J.M. and Bochner, F. (2000) Differential behavioral and neurochemical effects of para-methoxyamphetamine and 3,4- methylenedioxymethamphetamine in rat. Progress in Neuro-Psychopharmacology and Biological Psychiatry 24(6): 955-977.
|
|
Daws, L.C ., Gould G.G., Teicher, S.D., Gerhardt, G.A. and Frazer, A. (2000) 5-HT 1B receptor-mediated regulation of serotonin clearance in rat hippocampus in vivo. Journal of Neurochemistry75(5): 2113-2122.
|


